2. 上海交通大学 上海粒子物理和宇宙学重点实验室, 上海 200240;
3. 上海交通大学 致远学院, 上海 200240
2. Shanghai Key Laboratory for Particle Physics and Cosmology, Shanghai Jiao Tong University, 800 Dongchuan Road, Shanghai 200240, China;
3. Zhiyuan College, Shanghai Jiao Tong University, 800 Dongchuan Road, Shanghai 200240, China
化学工业中, 一个重要的过程是氢化反应(包括加氢和脱氢).完成这类反应, 几乎都要用到催化剂[1].一个熟悉的例子, 就是合成氨工业中用金属铂作为高效的多相催化剂[2].对于这类催化反应, 普遍的共识是, 和自由H原子不同, 晶体铂表面吸附的H原子受到周围铂离子作用, 性质发生改变, 从而化学活性明显增加.关于周边基质和表面H之间的作用机理, 文献[3]给出这一研究领域详细而深入的评论和总结, 重点讨论了H原子在固体表面上的化学吸附, 即H原子和基质原子/离子之间形成共价键.我们提出另外一种吸附H和金属表面相互作用的可能性.在很多情况下, 在晶体表面的近邻基质原子/离子所围成的凹陷位置上, 还稳定地存在很多填隙H原子, 它们没有和基质原子/离子形成共价键, 而是借助范德瓦尔斯静电力粘连在固体表面上(物理吸附).此时基质原子对这些填隙H原子主要施加泡利排斥作用(Pauli Repulsion), 从而改变H原子性质, 使其化学活性显著增加.我们将此作用简称为“固体表面填隙H的泡利激活”.这一新的催化机理可能和其他流行机制并存于表面.我们将说明, 新机制具有独特的优越性.很多情况下, 新催化机制可能有更大贡献.长期以来, 这种作用却没有受到应有的重视.此外, 对于给定的加氢反应, 如何选择有效的催化剂也是人们一直追求的目标.希望我们提出的新催化机制对这个问题的解决也会有所裨益.
泡利排斥是广泛存在于各类固体(包括纳米颗粒)表面的普遍作用.我们特别引进表面“泡利穴”(Pauli hole)的概念, 用以描写表面凹陷坑对外来填隙原子的泡利排斥作用, 便于定量地讨论/计算泡利斥力对这些填隙(或者替位式)原子性质的影响.重点讨论填隙H原子性质.为使读者了解我们的新观点, 简要介绍了我们已经发表过的工作[4].即列出“泡利穴”中填隙H原子满足的Schrödinger方程和边条件, 给出填隙H的基态波函数和基态能, 并与自由H原子做比较.以此说明, 受到泡利激活的填隙H性质已经明显不同于自由H.我们没有简单重复原有论文内容, 而是做了更深入的物理分析.着重指出填隙H两个主要的新特点:其基态电离能(代表其电子亲和力)显著小于自由H原子, 以及出现附加的诱导电矩.由此详细论证, 正是这两个物理特点, 使得填隙H不同于自由H, 其化学活性有了明显增加.此外, 还用这一新的化学激活机制解释一个近年令人困惑的问题:为什么过渡族金属“表面下的H原子”(subsurface H atom)在加氢反应中非常活跃, 具有很高的化学活性.而“表面H原子”(surface H atom)对加氢反应没有贡献[5-17].我们对此给出新的解释:实验中这种“表面下的H原子”, 就是被泡利激活的, 非常活泼的填隙H.
1 “泡利穴”(Pauli hole)泡利排斥的确广泛存在于各类固体表面上.只要表面凹陷坑尺寸适中, 和填隙原子(不局限于H)大小相匹配, 这种排斥力对填隙原子的影响就不可忽视.我们不讨论外来H原子的起源问题.实际上, 可以通过在固体周边H2分子气体中高频放电方法[18], 或者钨丝加热方法[19-21]获得原子H.对于过渡族金属, 情况更加简单:从周边到达固体表面的H2分子会自动解离, 成为H原子[22-26].
1.1 离子晶体这是最简单情况, 如食盐晶体NaCl. NaCl晶体为简单立方晶格, 如图 1所示.格点上的离子Na+, Cl-皆具有满壳层结构.在晶体表面, 每4个近邻离子(2个Na+和2个Cl-)围成一个四方形孔穴.置于四方孔穴中央低凹处的外来填隙原子的价电子将受到周边满壳层离子的泡利排斥. 图 1中红点表示填隙H或者其他填隙原子.这是穴中填隙原子(我们重点讨论H原子)的电子云受到的主要作用.在穴面整体为电中性的初级近似下, 可以忽略周边4个带电离子对填隙H原子的静电库仑力.泡利排斥是一种短程强作用, 只存在于满壳层原子球(图中为Na+, Cl-离子球)的表面上, 也可以说成图中四方穴的表面上.

|
图 1 NaCl晶体结构 Fig.1 The simple cubic structure of crystal NaCl (The green and yellow balls represent Cl- and Na+ ions with full shell structure, respectively. A smaller red ball, sited in central depression of a hole, formed by four adjacent Na+ and Cl- ions, represents a foreign interstitial H, which suffers a Pauli repulsive force from the surface of Na+ and Cl- ions. We simply call the square hole surrounded by adjacent Na+ and Cl- ions 'Pauli hole'.) |
把固体表面近邻基质原子(离子)围成的, 对中央填隙原子施加泡利排斥力的这类孔穴一律简称为“泡利穴”(Pauli hole).以下要说明, 它广泛存在于各类固体表面上, 不只限于离子晶体.
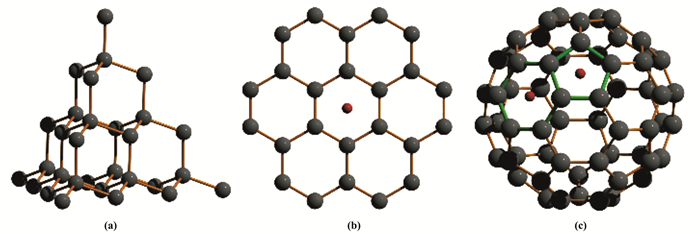
1.2 共价键晶体首先讨论由单一元素构成的单质共价键晶体, 如金刚石, 石墨, C60纳米球, Si单晶体等(见图 2).在其表面上, 任何一组近邻的基质原子围成的凹陷坑也是泡利穴.以石墨为例(图 2b), 其中相邻的C原子的键合力如C—C, C=C等皆为1共价键.其中每个C原子都是满壳层的.从而石墨表面近邻C原子围成的六边形孔洞成为泡利穴, 对穴中的填隙原子(图 2b中用红点表示一个填隙H原子)只施加泡利排斥力.
|
图 2 金刚石, 石墨以及C60的晶体结构 Fig.2 Examples of covalent monocrystals composed of a single element a. The structure of crystaldiamond(likewise for monocrystaline silicon; b. The graphene structure; c. The C60 nano-particle |
再说图 2c中的C60纳米球.它由60个C原子组成, 构成纳米球的60个顶点.球面上近邻的C原子共围成20个六边形和12个五边形, 分别为六边形和五边形泡利穴.它们对穴中填隙H原子(红点表示)都施加泡利排斥力.这里顺便提醒读者, H原子半径仅~0.05 nm, 明显小于石墨和C60六边形泡利穴的孔径尺寸, 并非最佳匹配.由此预期, 泡利排斥不会显著改变H性质.这些泡利穴或许更有利于改变其他尺寸较匹配的较大的填隙原子性质, 如锂(Li).
我们特别注意到, 图 2a的金刚石结构中, 它的任何一个晶面上, 近邻的C原子数目都比较多, 围成的孔穴几何尺寸都较大.所以对穴中原子几乎没有泡利排斥了.由于和填隙原子尺寸不匹配, 金刚石将不在讨论之列.
另一类是多质共价键晶体, 由两种以上元素构成.如SiO2, TiO2等.在SiO2单晶体中(图 3a), Si原子的排列和金刚石结构相同, 而O原子则套接在Si的晶格点阵中.其中每个O原子两侧都连接一个Si原子, 接近于直线排列, 形为Si—O—Si.其中的Si和O原子都是满壳层的.但与金刚石类似, SiO2单晶体中任何一个晶面上都不存在理想的泡利穴, 也是因为近邻的Si和O原子围成的孔穴几何尺寸太大.

|
图 3 a. SiO2(石英)单晶体结构; b.非晶态SiO2表面的各种泡利穴 Fig.3 a. The structure of crystal SiO2; b. Morphology of amorphous SiO2 (Note there are many Pauli holes with various shapes, e.g., triangles, squares, pentagonals, etc. All of them are surrounded by the adjacent Si and O atoms with full shell.) |
不过非晶态SiO2则是化学工业中经常采用的催化剂.非晶态SiO2结构中, 原子排列不再具有规则的空间周期性排列(图 3b).但近邻的满壳层Si和O原子围成各种小尺寸多边形泡利穴(如三边形, 四边形, 五边形等, 图 3b).对穴中填隙H有较强的排斥作用, 明显改变H或其他填隙原子性质. TiO2讨论从略.
1.3 金属晶体在金属表面, 近邻金属离子围成的表面凹陷坑也是理想的泡利穴.这一结论至少对碱金属和碱土金属正确.本节限于这类金属.过渡族金属则有必要在下节做更详细的讨论.
这类金属晶体中有两个主要成分, 其一是在格点上作空间周期性排列的金属离子, 它们为满壳层结构.另一成分是进入金属导带的价电子, 它们成为做公有化运动的自由电子.
众所周知, 金属中的自由电子并不是空间均匀分布的.其电子云密度分布也具有格点周期性.按照量子理论, 金属中自由电子的波函数形式为布洛赫函数, ψ(r, t) = U(r)exp(k·r-ωt), 波函数的振幅U(r)是周期函数, 具有与格点相同的周期性.在离子所在的格点位置处,振幅平方[U(r)]2最大, 表示每个格点离子周围自由电子密度最大, 这也符合我们的物理直观, 因为这些电子并非真正意义上的自由电子.在公有化运动中, 它受格点上带正电的金属离子库仑静电吸引, 会较长时间留在离子周围.所以, 布洛赫函数描写了金属自由电子运动的双重特性:既做公有化运动又较长时间处在离子束缚态上.指出金属自由电子云密度和离子有相同的空间周期性分布很重要:这样一来, 格点上每个正离子其实都被高密度自由电子云充分包围, 表观上接近电中性, 可近似看作“电中性的原”.从而把近邻金属离子围成的表面凹陷坑近似为电中性.这样, 在凹陷坑中, 填隙H受到的主要作用就只有泡利排斥了.首先, 金属的原子实是满壳层的, 它对填隙H原子只施加泡利斥力.其次, 自由电子是简并费米气体, 它对外来的填隙H原子施加费米简并压, 而费米简并压的本质就是泡利排斥.简言之, 金属表面近邻离子以及包围离子的自由电子云共同围成的凹陷坑确是“泡利穴”.
还要指出, 对于碱金属和碱土金属, 泡利排斥可能是表面H受到的唯一作用.其他的相互作用, 如H和表面基质原子的成键作用(金属—H键), 或者金属表面和填隙H之间的电子转移(电子交换)过程, 都难以存在.金属导带中的自由电子是非定域的, 由它和外来H难以形成’定域的’金属—H共价键, 这无需多做解释.自由电子和填隙H之间的电子转移(电子交换)同样不可能.对碱金属和碱土金属而言, 其费米能级都在EF~ -(2~3) eV范围中, 而填隙H的基态能级约为EH~-13.6 eV (暂不考虑和金属表面作用引起的修正), 远在费米能级之下.大的能级差表明, 填隙H原子基态电子不可能跃入费米能级之上成为导带中的自由电子.同样, 导带电子也不能进入H原子的1s态形成H-离子, 因为H-的基态能级为~ -0.8 eV, 明显高于金属费米能级EF~ -(2~3) eV.
1.4 化学吸附H覆盖层“表面下的泡利穴”由于过渡金属在催化的实际应用和理论探讨中很重要, 将给予更详细的讨论.论证的要点是, 过渡族金属表面同样存在“泡利穴”, 而且其“泡利穴”有新的特点:是表面化学吸附H覆盖层表面下的泡利穴.
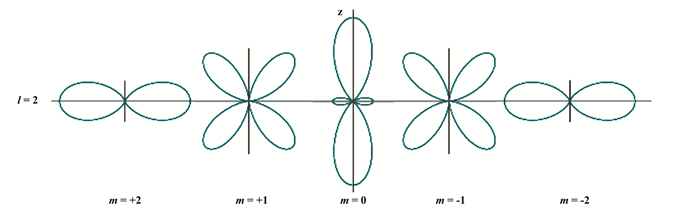
过渡族金属原子最外层为未填满的d壳层l=2或者f壳层(l=3).现以d壳层为例说明之. d壳层有5个可能轨道, 其轨道角动量的空间取向量子数分别为ml = 0, ±1, ±2.最多可容纳10个电子(即每个轨道可容纳自旋相反的两个电子). 图 4是d壳层l=2不同ml值的各态的电子云角分布示意图.图中的z轴在这里代表垂直于金属表面的方向.
|
图 4 d电子(l=2)各态的电子云角分布示意图 Fig.4 Angular distribution of the electronic cloud of quantum states of different ml values in d shell The Z axis is perpendicular to the surface of metals. |
由图 4看出, 过渡族元素金属原子, 的确不同于一般金属:其中量子数为(l=2, ml=0)的态, 原子符号为dz2, 它很特殊.其波函数有两个特点:
1) 电子波函数在核处(r=0)不是节点, 此处电子云密度ρe|r≠0.说明dz2态下, 原子核被高密度的电子云充分包围.在人们的一般印象中, 只有s态在核处才有不为零电子云密度, 其他态电子波函数在核处皆为零点. dz2态显然违反了这条规则, 使它明显不同于其他d态(见图 4).
2) 电子波函数只沿垂直表面方向(即图 4中的z轴方向)细细延伸.而其他d态波函数都在金属表面方向(水平方向), 或在靠近表面的方向延展(图 4).
这两个特点决定了过渡族金属表面态原子(离子)独特的性质.特点1表明, dz2态下, 电子与核之间的静电库仑力会比其他d态更强, 处于紧束缚状态.电子会长时间“定域”在表面的金属原子(离子)上, 其公有化运动较不明显.这一结论得到特点2的进一步支持.既然电子云沿垂直表面方向细长分布, 因此与其他d态电子不同, 相邻原子的dz2态电子波函数只有很少交叠, 暗示电子不易在相邻原子间自由转移.这再次表明, 处于dz2态的电子是近似归属于金属表面上的单个原子(离子)的, 可以忽略其公有化运动.简言之, 过渡族金属表面原子(离子)的dz2态实际上是定域态.
表面“定域态”的存在对过渡族金属表面性质至关重要.文献[3]所说H的化学吸附, 就是因为处于“定域态”dz2的表面金属原子(离子)的d电子和外来H原子的1s电子形成了共价键[27], 例如H—Pd键或H—Pt键等.而在碱金属和碱土金属表面, 只有满壳层的原子核和导带中的自由电子, 没有这类定域态电子, 很难形成定域的金属—H键.我们认为, 这可能是碱金属和碱土金属表面很难存在化学吸附H(参见文献[3])的原因.
正是上述表面H—Pd键或H—Pt键的存在, 帮助形成过渡族金属表面的“泡利穴”.如果化学吸附达到饱和(即几乎每个表面金属离子都和H形成共价键), 化学吸附H(成键的H)就会有高的表面覆盖率.以下说明, 此时形成的“泡利穴”是深埋在表面吸附H覆盖层下的.可以称为“表面下的泡利穴”.这是过渡族金属表面泡利穴的特点, 不同于其他固体.
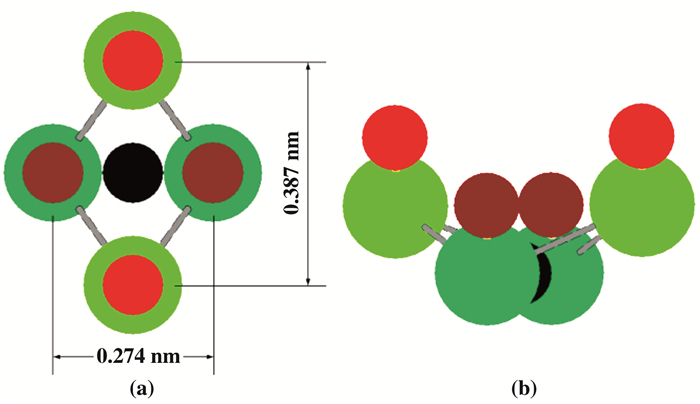
我们用单晶钯的(110)表面(当然不限于(110), 也可以是(111)面等)为例, 说明表面下的泡利穴的成因.钯是多相催化中广泛采用的催化剂, 在加氢反应中非常活泼[1].单晶钯是面心立方晶体(fcc), 在其(110)面上, 每四个近邻的钯原子就围成一个菱形凹陷槽, 如图 5所示(图 5a是(110)面的俯视图(正面图), 5b是(110)面的侧视图).它的尺寸恰好与H原子大小匹配, 是外来H的极佳填隙位置. 图 6是单个菱形凹槽图.大量的菱形凹槽依次毗连, 构成了钯的(110)面(见俯视图 5a).注意图 5和图 6中, 构成菱形凹槽的深绿和浅绿色球都表示钯离子, 不过两者不在同一平面上.深绿色球表示的钯离子, 其所在平面低于组成晶体(110)表面的浅绿钯离子0.137 nm.

|
图 5 单晶钯Pd的(110)面 Fig.5 The (110) crystal plane of metal Pd (Fig. 5 a. The front view of Pd (110) plane. Both the dark and the bright green balls represent the Pd ions. But they are not in the same plane, the dark green Pd ion is lower than the bright one ~0.137 nm. Note that in Fig. 5 a, every four adjacent Pd ions with different heights surround a rhomboid depression, suitable to arrange a foreign interstitial H(the smaller black ball). The smaller red balls in Fig. 5 a represent the chemisorbed H above (110) plane. If chemisorption reaches saturation, i.e., almost every Pd ion binds a adsorbed H, then there will be a H cover filled with chemisorbed H on (110) plane.Fig. 5 b. The side view of Pd (110) plane, where the relative positions of surface Pd ions to up chemisorbed H are clearer and obvious. The chemisorbed H (red ball) is higher than Pd ion(green ball) for about a H-Pd bond-length. The distance between dark green Pd ion and dark red H is the same. The black ball, sited in center of a rhomboid hole, represent interstitial H, with lower position than the chemisorbed H(the red and dark red ball)). |
|
图 6 钯单晶(110)面上的单个菱形泡利穴 Fig.6 The single rhomboid Pauli hole at Pd (110) plane (Fig. 6 a is front view of a single rhomboid hole. These four Pd ions, represented by green balls, are not in the same plane. The bright green ball is higher than dark one ~0.137 nm. Fig. 6 b is side view, which shows more obviously that, under the saturation adsorption, the rhomboid can be regard as a hole composed of 4 Pd ions and 4 chemisorbed H). |
到此为止, 这些菱形凹陷槽还不是“泡利穴”, 因为这4个近邻钯离子外层都不是满壳层的.详细说明如下:已经知道, 对于自由的钯原子而言, 其最外面的4d壳层已经填满10个电子, 可见它原本是满壳层的(自由钯原子的最外层n=4的电子组态是4s24p64d10, 已经填满18个电子).对应于最外层4 dz2态的两个填充电子, 其第一和第二电离能分别为~ 7 eV和~19 eV.即对应的基态能级分别为E1≈7 eV和E2≈-19 eV.但是, 晶体表面的钯离子不同于自由钯原子, 会受到自由电子气的费米简并压(本质是泡利排斥作用)扰动.换言之, 每个表面钯离子其实处在自由电子气构成的泡利穴中.其能级会提高.它的第一基态能级由~ 7 eV上升到≥-5 eV, 已经高于钯晶体的费米能级EF≈-5.12 eV了.其后果是表面钯4dz2子壳层不再是被两个电子同时占有的满壳层, 因为其中的一个电子会向导带转移.这是和自由钯原子不同的新特点(但是能级提升后, 表面钯的第二电离能仍然很大, 第二个电子不会电离, 表面4dz2束缚态稳定在单电子态上,即表面钯成为Pd+.
但当单晶钯周边充满H2分子气体时, 情况发生很大变化.到达固体表面的H2分子会自动解离, 成为H原子.其后是这些H原子的化学吸附[22-26].所形成的H—Pd共价键使吸附H的基态1s和Pd的4 dz2定域态因共用电子对而同时成为满壳层.所以, 在H饱和吸附时, 菱形凹陷穴实际上由4个近邻的H—Pd共价键围成(见图 6b, 它包括了上面4个H和下面4个钯).其中的H和Pd都已经满壳层, 遂使菱形穴成为理想的“泡利穴”.穴的高度约为H—Pd共价键的键长. 图 5, 6中, 红色及暗红色实心球都代表H—Pd共价键上的表面H原子, 但暗红H原子是连接在深绿钯离子上的, 所以其高度也相应地比亮红色H原子低~ 0.137 nm.
我们强调, 对于因物理吸附而黏贴在泡利穴底部的填隙H而言(图 6b中用黑球表示), 主要施加泡利排斥作用的, 其实是穴底部的4个钯原子, 因为它们才是紧密环绕黏贴在底部填隙H的最近邻.相比之下, 上方4个成键H原子对填隙H的泡利作用很弱, 可以忽略不计.
可见, 过渡金属Pd(或Pt)有不同于其他固体的特点:它是借助化学吸附H的Pd—H共价键才成为泡利穴的.其泡利穴比较深, 深度约为一个Pd—H键长.深埋于表面最上方化学吸附H(图中用红和暗红球表示)的覆盖层之下.因此, 黏贴在泡利穴底部的填隙H(图 5, 6中的黑球)应当称为“表面下的H原子”(subsurface H atom), 围绕它的4个Pd+离子组成的孔穴应当称为表面下的泡利穴.在最上方吸附H覆盖层中, 那些用红色(及暗红色)圆球表示的成键H原子才是“表面H原子”(surface H atom).今后在讨论过渡族金属催化作用时, 我们将特别区分“表面H原子”和“表面下方”泡利穴中的填隙H原子.在以下的章节将说明, 覆盖层下方泡利穴中的填隙H的性质和Pd—H键上的表面H非常不同, 它在化学反应中表现更加活跃.
在各类固体的表面, 包括碱和碱土金属, 过渡族金属, 共价键晶体(晶态和非晶态, 单质和多质), 离子晶体等等, 广泛存在表面基质原子(离子)对外来填隙H原子的泡利排斥.我们把固体表面安置填隙H的凹陷坑称为“泡利穴”.其实泡利穴不仅存在于固体表面上,在均相催化中, 在溶液中经常采用苯或二甲苯一类的催化剂.它们也有类似的泡利穴结构.构成苯分子的6个C原子由共价键彼此相接, 围成苯环.其中每个C原子都具有满壳层结构.因此苯环就是一个六边形“泡利穴”.置于其中央的外来原子(如H, Li, C或O等), 同样受到周边满壳层C原子的泡利排斥.这种泡利穴由近邻的基质原子(离子)围成.这些近邻原子(离子)都具有满壳层结构.我们在以下章节中将证明, 位于泡利穴中的外来填隙H原子会在泡利斥力作用下改变其物理性质, 使其化学活性明显增加.
2 泡利穴中填隙H原子 2.1 Schrödinger方程和边条件为理解固体表面的泡利排斥如何改变吸附H原子的性质, 我们曾经求解过“泡利穴”中填隙H原子的Schrödinger方程, 并由此得到穴中填隙H原子的基态波函数Φ和基态能量E(参见文献[4]).我们曾将它和自由H原子基态做比较, 的确看到有明显的改变.从这一改变了的基态的(Φ, E), 可以阐明穴中填隙H在泡利斥力约束下化学活性的增加。所得到的解对研究催化问题可能具有潜在重要性.此外, 利用这一改变了的波函数Φ还可以解释, 外来H原子一旦进入填隙位置后, 为什么就会稳定地粘连在固体表面上, 成为真实的物理存在.详细推导和讨论参见我们以前的论文(Phy. Rev. B, 41, 8180, 1990, You and Ye)[4].论文全名是:“固体表面填隙式或替代式H原子性质”(‘Properties of a substitutional and/or interstitial surface hydrogen atom’).其中提到的“替代式H原子”指的是固体表面格点中的一个基质原子(或离子)被外来“杂质”H原子所替代.填隙式和替代式H原子的共同特点是它们都被近邻的满壳层基质原子(离子)所包围, 都处在“泡利穴”中, 数学上可以一并处理.
为了方便新催化机制的讨论, 对该文做一个简短的概括性回顾很有必要:首先, 尽管各种固体表面上的“泡利穴”形状大小各异, 但为了便于求得解析解, 我们在该文中用一个绕Z轴的, 形状是旋转抛物面的凹陷坑简单地描写“泡利穴”, 如图 7所示(不用半球面近似, 因为它没有解析解).图中, 固体表面设为X-Y坐标面, Z轴垂直表面.该旋转抛物面的焦点F就安置在固体表面(X-Y平面)上, 并将它选为坐标原点.旋转抛物面和固体表面截出一个园, 圆心就在点F, 半径记为ξ0, 文中ξ0是文中以玻尔半径a0为单位的无量纲半径, 代表“泡利穴”的大小.图中PF=ξ0/2是抛物面的焦距, 代表“泡利穴”的深度.计算中, 表面填隙H原子的核就安置在原点F.对于抛物面, 采用抛物线坐标(ξ, η, z)代替笛卡尔坐标(x, y, z)或球坐标(r, θ, ϕ)是最方便的[4].所得电子波函数解以抛物坐标为自变量, 为Ф=Ф(ξ, η, z).描写旋转抛物面的抛物线坐标和笛卡尔坐标(x, y, z)之间的关系为:

|
图 7 固体表面“泡利穴”的简化模型 Fig.7 The simplified model of the Pauli hole at surface of solids (The Pauli hole is represented by a paraboloid of revolution around Z axis. The regularly arranged black spots represent the substrate atoms/ions sited at lattice points of the crystal. the X-Y coordinate plane is arranged on the surface of solids. Z-axis is perpendicular to solid surface. The focus F of paraboloid of revolution around Z-axis is put at this surface. The nucleus of H atom is put at F point. The section of paraboloid with solid surface is a circle with radius ξ0 (ξ0 is in the unit of Bohr radius a0). The center of circle is at focus F. PF= ξ0/2 is the focal length of paraboloid, represents the depth of Pauli hole. The electronic cloud of interstitial H can not penetrate into the solid through the paraboloidal wall due to the Pauli repulsion. This fact can be expressed by a boundary condition which the electronic wave function Φ of H has to satisfy. Namely, at the paraboloidal wall ξ=ξ0 (the surface of Pauli hole), Φ(ξ0, η, z)=0). |
| $ x=\sqrt{\xi, \eta} \cos \varphi, y=\sqrt{\xi, \eta} \sin \varphi, z=(\eta-\xi) / 2 $ | (1) |
ξ和η取值范围是(0, ∞), φ为(0, 2 π).由(1)式不难看出, 当ξ为常数时(即(ξ=ξ0), 且ξ0>0), 就得到一个绕Z轴向上开放的旋转抛物面.
泡利穴中的填隙H原子的Schrödinger方程以及波函数满足的边条件分别为:
| $ \hat{H} \varPhi=E \varPhi, \varPhi\left(\xi_{0}, \eta, z\right)=0\\ \left.\Phi\left(\xi_{0}, \eta, z\right)\right|_{\eta \rightarrow \infty}=0, (自然边界条件) $ | (2) |
由于泡利排斥作用, 填隙H原子的电子云无法穿过抛物面(可称为“泡利墙”)渗透到固体内部, 它只能约束在抛物形“泡利穴”表面以内.这一事实就用上述边条件表示, 即填隙H原子的电子波函数Φ在“泡利穴”表面上(即旋转抛物面ξ=ξ0上)必须为零.由于穴表面对填隙H的泡利排斥作用已经用边条件充分表达了, 不必在哈密顿量中再增加表示泡利作用的附加项.因此, (2)式中仍然采用自由H原子的哈密顿量, 即:
| $ \hat{H}=\frac{\hbar^{2}}{2 m_{0}} \nabla^{2}-\frac{e^{2}}{r} $ | (3) |
以下再简要说明方程(2)的基态解(Φ, E)的性质(讨论催化问题主要涉及基态), 指出它不同于自由H原子基态(1s态)的主要点.
首先讨论基态波函数Φ的改变.该解析解Φ用超越几何函数表达.没有必要列出波函数详细的数学表式及其推导(详见文献[4]), 仅用图 8示出填隙H原子基态波函数给出的电子云密度空间分布(即[Ф]2分布)等高线图已经足够了.比较图 8a, b可见, 在排斥作用下, 填隙H原子的基态电子云密度分布特征的确不同于自由H原子的1s态.它已经变形, 不再是球对称分布, 而是沿着垂直固体表面方向向上延伸拉长.由于电子云的拉伸形变, 填隙H的负电中心不再与正电中心重合, 诱导出一个电矩p, 它垂直固体表面并指向固体内部.
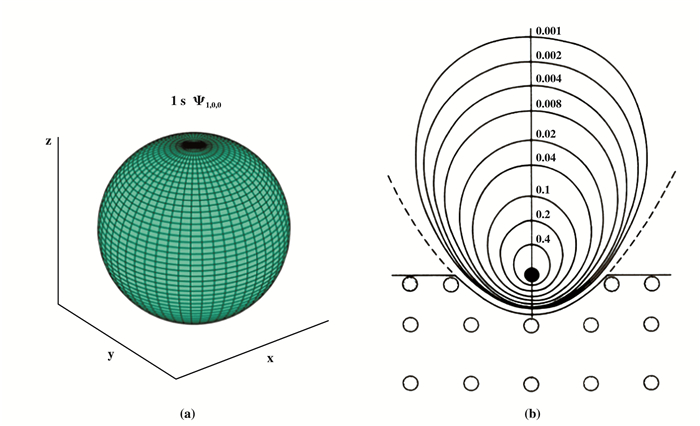
|
图 8 自由H原子基态1s的球对称电子云分布 Fig.8 The spherically symmetric distribution of electronic cloud of ground state 1s of a free H (Fig. 8 b. The contours of probability density(|Ф|2) of the ground state of interstitial H(taken from [4]), confined in a Pauli hole with a simplified Pauli wall of paraboloid of revolution. The black point represents the nuclear of H, the hollow circle represents the substrate atoms with closed shell. The electronic cloud of confined H atom no longer has a distribution of spherical symmetry. The deformation of wave function is obvious, which is elongated along the direction perpendicular to the solid surface, thus induced an electric dipole p, vertically points to the interior of solid.) |
其次, 填隙H的基态能量E也相应发生改变, 如图 9和表 1(均取自[4])所示. 图 9给出泡利穴中填隙H的诱导电矩p和基态能量E随泡利穴的半径ξ0的变化曲线p ~ ξ0和E ~ ξ0. 表 1是图 9中曲线的数值表示, 只是表中把基态能量E换成应用上更加方便的基态电离能I.两者关系为I=-E, 只差一个负号, E < 0而I>0.电离能I比基态能E更具物理直观性, I值代表电子亲和力大小.因此我们也常用基态电离能I代替基态能级E, 如表 1所示.
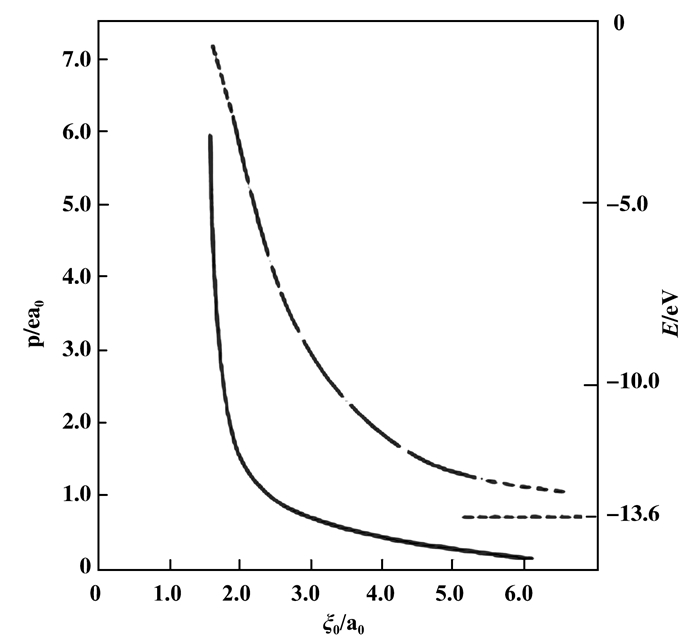
|
图 9 填隙H的诱导电矩p (单位是ea0)和基态能量E(eV)随泡利穴半径ξ0 Fig.9 The variations of induced dipole p (in unit ea0) and energy of ground state E(in unit eV) with the radius of Pauli hole, p ~ ξ0 and E ~ ξ0 (quoted from [4]). ξ0 is a dimensionless radius in unit a0, the Bohr radius. From Figure 9 we see, both the dipole p and the energy E(E < 0) are monotonically increased with decreased radius ξ0. In fact, ξ0 scales the strength of Pauli repulsion. |
| Table 1 Numerical relations of the ionization potential I (I>0) (induced dipole p of gound state of confined H with Pauli radius ξ0 (quoted from [4]). Table 1 is the numerical representation of Fig. 9.) |
图 9和表 1都说明, 在泡利斥力约束下, 填隙H基态能量EH(EH < 0)明显提升, 高于自由H原子, 即EH> -13.6 eV(以下, 凡属描写填隙H原子性质的物理量皆用下脚标H标记, 以区别参与化学反应的其它原子).或者等价地, 其基态电离能IH明显小于自由H, 即IH < 13.6 eV.这表示, 比起自由H, 泡利斥力作用下的填隙H变得更容易电离(即填隙H的电子亲和力明显下降), 更易与外来原子交换电子.电离能IH的减小表明其化学活性增大.
图 9和表 1还表明, ‘泡利穴’的半径ξ0是影响填隙H性质的重要参量.孔穴半径ξ0大小在物理上表示泡利约束作用之强弱.半径ξ0越小, 显然排斥作用越强, 被挤压的电子云(见图 8b)向上方自由空间的扩展就越大.相应地, 电子到氢核H+的平均距离r=∫Φ*rΦdτ也越大, 表示电子更远离氢核.这导致诱导电矩PH=er增大, 同时也导致电子与H核之间静电库仑引力减弱, 使电离能IH减小.由此可见, 电离能IH的减小和电矩PH的出现, 原来都是由填隙H基态波函数的形变引起的, ΦH, IH和PH, 三者关系紧密.
在此附带说明, 既然泡利穴半径ξ0是决定作用强弱的重要参量, 所以在选择催化材料时, 首先就是考量该孔穴半径值.各种固体材料, 其表面“泡利穴”的形状, 大小都不相同.同一晶体在不同晶面上的孔穴大小也不相同.它们的实际形状也复杂多样, 和简化的旋转抛物面很不同.在晶体表面上的截线当然也不会是简单的圆形.为了把图 9和表 1近似地应用于实际固体催化剂做半定量计算, 它在讨论具体的化学反应时, 至少在半定量分析中十分有用.
以上就是对我们前文[4]的简短概述.我们进一步认识到, 其实填隙H的诱导电矩PH同样重要, 它和电离能IH的减小一样, 深刻影响填隙H原子的化学活性,是讨论催化问题中不可忽略的重要因素.至少, 电矩PH的以下4个功效和催化密切相关:
1.它保证填隙H原子稳定地黏贴在“泡利穴”中.如上所述, 电矩PH垂直表面并指向固体内部.它必然在晶体表面感应一个带负电的面电荷分布(对金属, 是自由电子面电荷分布, 对固体介质, 为极化面电荷), 感应面电荷与电矩PH之间的静电吸引使外来填隙H原子“瞬时地粘黏”于表面.所以, 我们讨论的填隙H原子, 是物理吸附于固体表面的稳定的物理存在, 而不是一个假想的模型考虑.
2.电矩PH的偶极电场对周边环境中的正离子(如N++, O+等)的静电吸引和对中性原子分子的范德瓦斯吸力, 会使外来原子(离子)有沿PH的电场线移动的倾向, 造成对填隙H原子的靶向轰击.填隙H成为靶原子.显然相比气体中的随机碰撞而言, 这种靶向轰击增大了外来原子对H的碰撞概率, 对加氢反应更为有利.
3. 表 1中, 电矩PH足够大, 其电场会使周边气体中的靠近填隙H的原子或离子发生明显拉伸形变, 自然造成这些外来原子电离能IX减小(以下, 外来原子一概用X来标记).它们在向填隙H原子接近并最终碰撞的过程中, 伴随原子拉伸形变而逐渐减小的电离能IX会不断与填隙H电离能IH趋同, 即IX→IH(注意IH已经很小.只要ξ0取较小值, 就有IH ≪ 13.6 eV, 见表 1).最终近似相等, IX≈IH (也可以等价地表示为基态能量EX≈EH).限于篇幅, 我们将在另文中对趋同, 近似相等的必然性做更深入的, 半定量的物理论证.顺便指出, 在此过程中, 填隙H的电离能几乎不变, IH ≈Const(同样在另文中证明).显然, 两个原子接近时的’能量趋同’效应, 有利于共价键形成.只当不断靠近的外来原子和填隙H原子的两个原子波函数Ψ1和Ψ2最终成了’同一能级’上的’简并态’, 交叠的原子波函数才可以通过线性组合, 成为分子波函数Ψ12=C1Φ1+C2Φ2 (即分子轨道[28]).所以, 能级趋同, 产生简并, 是形成分子波函数应该满足的前提条件.这也符合人们的物理直观:能量趋同保证了两个原子的价电子可以在原子间无能量障碍地自由迁移, 成为共用电子, 从而形成共价键.
4.填隙H强的非零电矩PH造成外来原子X形变,也诱导出一个非零电矩PX.电矩PX沿PH的电场EH取向, 即PX‖EH.最终, 发生碰撞时, PX将与填隙H电矩PH平行, PX‖PH(因为在填隙H处必有EH‖PH. PX‖PH对成键至关重要:在很多情况下, 它使得两个交叠原子波函数固有的空间对称性(这种对称性在量子力学中称为’宇称’)彼此适配.只有这样, 才不会出现两个原子碰撞时, 相互叠加的原子驻波态Ψ1和Ψ2产生干涉相消的情况.以此保证共价键得以形成. PX‖PH通常就是理想的’最佳碰撞方位’.限于篇幅, 我们将在另文中对“宇称匹配”, 避免干涉相消对形成共价键的重要意义给与详细的说明.
总之, 固体表面的泡利排斥作用会深刻影响填隙H原子性质, 明显增加了其化学活性.我们将此催化作用简称为’填隙H的泡利激活’.
3 结论和讨论泡利排斥作用是广泛存在于各类固体表面(包括纳米颗粒表面)的作用力.它使得填隙H的性质不同于自由H原子, 其化学活性明显增加, 在氢化反应中显得非常活跃.我们把这种催化效应简单称为“填隙H原子的泡利激活”.填隙H化学活性的增加和它的两个新特点密切相关:即其电离能IH的显著降低(电子亲和力下降), 以及非零电矩PH的出现.两者缺一不可.
这是氢化反应(加氢)中一种过去被长期忽视的催化机理, 值得深入研究.很有可能, “泡利激活”是已有的催化理论的一个重要补充.尤其对加氢反应中经常采用的过渡族金属催化剂(Pt, Pd, Fe, Ni等等)而言, 除了要考虑以共价键连接基质离子的“表面H原子”之外(例如H—Pd或H—Pt键上的H,即文献[3]中所说的“化学吸附H”), 也不能忽视数量很大的, 吸附在各个泡利穴底部的“表面下的填隙H原子”(物理吸附), 它们应当受到同等关注.在一些情况下, 后者的催化作用可能更加重要.
近年的很多报道中, 对“表面下的H原子”在氢化反应中的重要贡献都给于明确的肯定.而“表面H原子”的化学活性反而没有得到实验证实[5-17].已经有不少相应的理论探讨, 但都没有涉及我们说到的填隙H的泡利激活.我们认为, 过渡金属“深埋在表面下的泡利穴”中的, 被泡利激活的填隙H, 其实就是他们实验中的“表面下的H原子”(subsurface H atom), 它们在化学反应中非常活跃.这些填隙H的位置显著低于成键的“表面H原子”(surface H atom). 图 5和图 6清楚显示了这两种H原子的相对高度.
毋庸置疑, “泡利穴”中的填隙H应当更具化学亲和性.它是物理吸附的, 所以基态尚未填满.从而易与外来原子(离子)共用电子对, 形成共价键.与此相反, 对于已经成键的(如H—Pd或H—Pt), 化学吸附的“表面H原子”, 基态已具有稳定的满壳层结构, 不利于加氢反应.但是我们强调, 填隙H原子化学活性增加的更重要原因仍然是泡利激活.泡利排斥使填隙H基态波函数发生了改变.受挤压的电子云在垂直表面的自由空间方向向上延申, 其舌尖实际上已经冲出表面H的覆盖层(参考图 6a, b), 自然有利于填隙H和气体中的外来原子X接触, 促使X—H共价键的形成.但是这一几何因素也不是最重要的.泡利排斥造成的波函数形变, 使其电离能IH明显减小和非零诱导电矩PH出现, 才是填隙H化学活性增加最重要的原因.
我们对新催化机制的认识仅仅停留在定性物理讨论水平上, 缺少定量性, 但是这些特点已经鼓励了我们, 把更多注意力聚焦于“表面下的H原子”——泡利穴中的填隙H.最重要的任务, 是要把填隙H电矩PH四大贡献中关键的后两个, 即两个原子在接近过程中能量趋同及其波函数的空间对称性适配, 对它做更详细的, 物理上更容易理解的半定量论证.我们将在后续的论文中完成这一论证.
把填隙H诱导电矩PH最后两个效应(第3和第4条)说得清楚很重要, 因为它们与形成共价键应满足的两个前提条件密切相关.我们将在后续论文中, 首先详细说明什么是形成共价键的前提条件.然后在半定量水平上论证, 泡利激活后的填隙H, 借助于强的非零诱导电矩PH电场力的作用, 恰好能使这些前提条件得到满足, 从而促进了共价键形成.这无疑是泡利激活机制的独特优越性.
我们主要讨论多相催化问题(固体表面填隙H原子的催化).但是“泡利激活”原则上可以推广到均相催化中.因为在均相催化中经常使用的催化剂通常也具有类似的泡利穴结构.
我们只讨论了泡利穴中填隙H的催化.但是原则上不难推广到其他元素.我们计划用类似方法探讨石墨烯表面填隙锂原子的泡利激活.
近来的天文观测中发现, 很多有机分子云团中(例如H2O, CH4, C2H2, C2H4, …等云团), 同时存在一些尺寸约0.001~10 μm的尘埃物质(如C颗粒, SiO2颗粒等等).两者的并存使我们猜测, 或许这些尘埃物质(包括纳米颗粒)本身就是多相催化剂, 其表面存在的“泡利穴”可能对天文分子的形成有重要贡献.
致谢: 作者十分感谢上海交通大学应用化学系李新昊和宰建陶二位教授有益的讨论和帮助, 使这项研究得以顺利完成.中国科学技术大学物理系程富华教授,化学系黄复华教授也提供了必要的帮助, 在此一并感谢!
| [1] |
Peter Ferrin, Shampa Kandoi, Anand Udaykumar Nilekar, et al. Hydrogen adsorption, absorption and diffusion on and in transition metal surfaces: A DFT study[J]. Surf Sci, 2012, 606(7): 679–689.
|
| [2] |
Ertl G, Knozinger H, Weitkamp J. Handbook of heterogeneous catalysis[M]. Weinheim: Wiley-VCH, 1997.
|
| [3] |
Christmann K. Interaction of hydrogen with solid surfaces[J]. Surf Sci Rep, 1988, 9(1): 1–163.
|
| [4] |
YouJ H, Ye Z G, Du M L. Properties of a substitutional and/or interstitial surface hydrogen atom[J]. Phys Rev B, 1990, 41(12): 8180–8183.
DOI:10.1103/PhysRevB.41.8180 |
| [5] |
JohnsonA D, Daley S P, Utz A L, et al. The chemistry of bulk hydrogen: Reaction of hydrogen embedded in nickel with adsorbed CH3[J]. Science, 1992, 257(5067): 223–225.
DOI:10.1126/science.257.5067.223 |
| [6] |
Ceyer S T. The unique chemistry of hydrogen beneath the surface: catalytic hydrogenation of hydrocarbons[J]. Acc Chem Res, 2001, 34(9): 737–744.
DOI:10.1021/ar970030f |
| [7] |
Teschner D, Borsodi J, Wootsch A, et al. The roles of subsurface carbon and hydrogen in palladium-catalyzed alkyne hydrogenation[J]. Science, 2008, 320(5872): 86–89.
DOI:10.1126/science.1155200 |
| [8] |
Henkelman G, Arnaldsson A, Jonsson H. Theoretical calculations of CH4 and H2 associative desorption from Ni(111): Could subsurface hydrogen play an important role?[J]. J Chem Phys, 2006, 124(4): 044706–044706.
DOI:10.1063/1.2161193 |
| [9] |
Welde M, Fukutani K, Ludwig W, et al. Influence of carbon deposition on the hydrogen distribution in Pd nanoparticles and their reactivity in olefin hydrogenation[J]. Angew Chem Int Edit, 2008, 47(48): 9289–9293.
DOI:10.1002/anie.200801923 |
| [10] |
a. Freund H J. Influence of carbon deposition on the hydrogen distribution in Pd nanoparticles and their reactivity in olefin hydrogenation[J]. Angew Chem Inter Edit, 2008, 47(48): 9289-9293. b. Freund H J. Einfluss von kohlenstoffablagerungen auf die wasserstoffverteilung in Pd-nanopartikeln und deren reaktivität in der olefinhydrierung [J]. Angew Chem, 2008, 120(48): 9430-9434. |
| [11] |
a. Doyle A M, Shaikhutdinov S K, Jackson S D, et al. Hydrierung an metalloberflachen: Warum sind nanoteilchen aktiver als einkristalle? [J]. Angew Chem Int Edit, 2003, 115(42): 5240-5243. b. Doyle A M, Shaikhutdinov S K, Jackson S D, et al. Hydrogenation on metal surfaces: Why are nanoparticles more active than single crystals? [J]. Angew Chem, 2003, 115(42): 5398-5401. |
| [12] |
Ludwig W, Savara A, Dostert K H, et al. Olefin hydrogenation on Pd model supported catalysts: New mechanistic insights[J]. J Catal, 2011, 284(2): 148–156.
DOI:10.1016/j.jcat.2011.10.010 |
| [13] |
Ludwig W, Savara A, Strauermann S, et al. Role of low-coordinated surface sites in olefin hydrogenation: A molecular beam study on Pd nanoparticles and Pd(111)[J]. Chem Phys Chem, 2010, 11(11): 2319–2322.
DOI:10.1002/cphc.201000355 |
| [14] |
Armbrüster M, Behrens M, Cinquini F, et al. How to control the selectivity of palladium-based catalysts in hydrogenation reactions: The role of subsurface chemistry[J]. Chem Cat Chem, 2012, 4(8): 1048–1063.
|
| [15] |
Ledentu V, Dong W, Sautet P. Heterogeneous catalysis through subsurface sites[J]. J Am Chem Soc, 2000, 122(8): 1796–1801.
DOI:10.1021/ja983975g |
| [16] |
Michaelides A, Hu P, Alavi A. Physical origin of the high reactivity of subsurface hydrogen in catalytic hydrogenation[J]. J Chem Phys, 1999, 111(4): 1343–1345.
DOI:10.1063/1.479392 |
| [17] |
Aleksandrov H A, Vines F, Ludwig W, et al. Tuning the surface chemistry of Pd by atomic C and H: A microscopic picture[J]. Chem Eur J, 2013, 19(4): 1335–1345.
DOI:10.1002/chem.201201106 |
| [18] |
Haberrecker K, Mollwo E, Schreiber H, et al. The ZnO-crystal as sensitive and selective detector for atomic hydrogen beams[J]. Nucl Instr Methods, 1967, 57(1): 22–28.
|
| [19] |
Langmuir I. A chemically active modification of hydrogen[J]. J Am Chem Soc, 1912, 34(10): 1310–1321.
|
| [20] |
Langmuir I, Mackey G M J. The dissociation of hydrogen into atoms. Part Ⅰ. Experimental[J]. J Am Chem Soc, 1914, 36(8): 1708–1720.
DOI:10.1021/ja02185a011 |
| [21] |
Hickmott T W. Interaction of hydrogen with tungsten[J]. J Chem Phys, 1960, 32(3): 810–823.
DOI:10.1063/1.1730802 |
| [22] |
Gregory A, Gelb A, Silbey R. A simple quantum chemical theory of dissociative adsorption[J]. Surf Sci, 1978, 74(2): 497–523.
DOI:10.1016/0039-6028(78)90041-9 |
| [23] |
Harris J, Anderson S. H2 dissociation at metal surfaces[J]. Phys Rev Lett, 1985, 55(15): 1583–1586.
DOI:10.1103/PhysRevLett.55.1583 |
| [24] |
Poelsema B, Palmer R L, Mechterscheimer G, et al. Helium scattering as a probe of the clean and adsorbate covered Pt(111) surface[J]. Surf Sci, 1982, 117(1): 60–66.
|
| [25] |
Norskov J K, Houmoller A, Johansson P K, et al. Adsorption and dissociation of H2 on Mg surfaces[J]. Phys Rev Lett, 1981, 46(4): 257–260.
DOI:10.1103/PhysRevLett.46.257 |
| [26] |
Bond G C. Metal-catalysed reaction of hydrocarbon[M]. New York: Springer, 2005.
|
| [27] |
Nordlander P, Holloway S, Norskov J K. Hydrogen adsorption on metal surfaces[J]. Surf Sci, 1984, 136(1): 59–81.
DOI:10.1016/0039-6028(84)90655-1 |
| [28] |
Olive G H, Olive S. Coordination and catalysis[M]. Verlag Chemie, 1977.
|
| [29] |
Heitler W, London F. Wechselwirkung neutraler atome und homoopolare bindung nach der quantenmechanik[J]. Zeits F Physik, 1927, 44(6): 455–472.
|
| [30] |
Zeng Jin-yan(曾谨言). Quantum mechanics(量子力学)[M]. Beijing: Science Press(科学出版社), 1981.
|
| [31] |
Zhou Shi-xun(周世勋). A course of quantum mechanics(量子力学教程)[M]. Beijing: People's Education Press(人民教育出版社), 1979.
|
| [32] |
Voldenstein M B (伏尔坚斯坦), Wang Ding-chang(王鼎昌). Molecular Optics(分子光学)[M]. Beijing: Higer Education Press(高等教育出版社), 1958.
|
| [33] |
Born M, Wolf E. Principles of Optics[M]. Ox-ford: Pergamon, 1970.
|
 2019, Vol. 33
2019, Vol. 33


